- Blog
- Order of atomic orbitals
- Voice ringtones free download
- Patina green copper hair colors
- Shiira momo
- 500 watt halogen bulb
- Suburbia game youtube
- Juegos de intruder combat training 2 hacked
- Team sonic racing overdrive videos
- Dungeon hunter 4 mod apk revdl
- Coupon for batch pdf merger
- Image vectorizer for gran turismo sport
- Viscosity for mac
- Softperfect wifi guard win 8-1
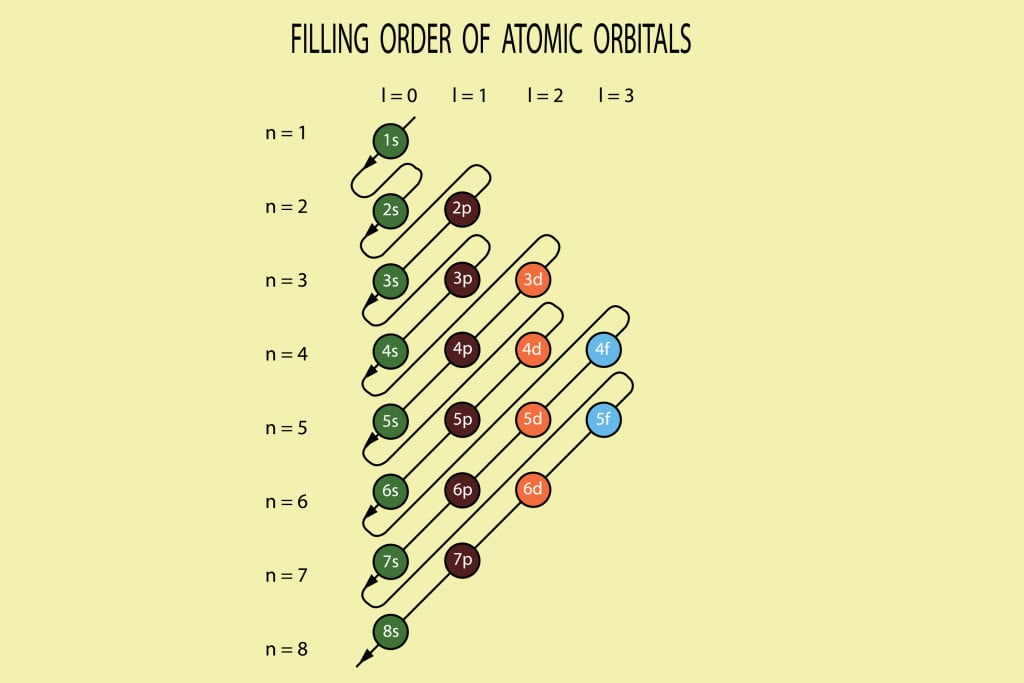
Dalton proposed that every matter is composed of atoms that are indivisible and. Primarily, the atomic structure of matter is made up of protons, electrons and neutrons. When electrons fill the energy levels, it fills principal energy levels, sublevels, atomic orbitals from lowest energy first. Ascending order of energy 1s, 2s, 2p, 3s, 3p, 4s, 3d.

The distance between the nucleus and the outer shell increases. The energy of orbital (p + l) value it two orbitals have same (n + l) value, E n. As the value of n increases (Principal quantum number), energies of the orbitals increases. Below is a simple scheme to help remember the order in which the orbitals are filled. Atomic Structure: The atomic structure of an element refers to the constitution of its nucleus and the arrangement of the electrons around it. Aufbau’s principle: The filling of electrons should take place in accordance with the ascending order of energy of orbitals: Lower energy orbital should be filled first and higher energy levels.
\( \newcommand\): Since the core electron shells correspond to noble gas electron configurations, we can abbreviate electron configurations by writing the noble gas that matches the core electron configuration, along with the valence electrons in a condensed format. Electrons in an atom are grouped around the nucleus into shells.
- Blog
- Order of atomic orbitals
- Voice ringtones free download
- Patina green copper hair colors
- Shiira momo
- 500 watt halogen bulb
- Suburbia game youtube
- Juegos de intruder combat training 2 hacked
- Team sonic racing overdrive videos
- Dungeon hunter 4 mod apk revdl
- Coupon for batch pdf merger
- Image vectorizer for gran turismo sport
- Viscosity for mac
- Softperfect wifi guard win 8-1
